
Pembrolizumab prolonged OS versus chemotherapy as second-line therapy for advanced esophageal cancer in patients with PD-L1 CPS ≥ 10, with fewer treatment-related adverse events. Grade 3-5 treatment-related adverse events occurred in 18.2% of patients with pembrolizumab versus 40.9% in those who underwent chemotherapy. 0095) in patients with squamous cell carcinoma and 7.1 months versus 7.1 months (HR, 0.89 P =. Median OS was 8.2 months versus 7.1 months (HR, 0.78 P =. Primary end points were overall survival (OS) in patients with programmed death ligand-1 (PD-L1) combined positive score (CPS) ≥ 10, in patients with squamous cell carcinoma, and in all patients (one-sided α 0.9%, 0.8%, and 0.8%, respectively).Īt final analysis, conducted 16 months after the last patient was randomly assigned, OS was prolonged with pembrolizumab versus chemotherapy for patients with CPS ≥ 10 (median, 9.3 v 6.7 months hazard ratio, 0.69 P =. In this open-label, phase III study, we randomly assigned (1:1) 628 patients with advanced/metastatic squamous cell carcinoma or adenocarcinoma of the esophagus, that progressed after one prior therapy, to pembrolizumab 200 mg every 3 weeks for up to 2 years or chemotherapy (investigator's choice of paclitaxel, docetaxel, or irinotecan). FDA also approved the PD-L1 IHC 22C3 pharmDx (Dako North America, Inc.) as a companion diagnostic for selecting patients with TNBC for pembrolizumab. Patients with advanced esophageal cancer have a poor prognosis and limited treatment options after first-line chemotherapy. 24 CHU Brest - Institut de Cancerologie et d'Hematologie, Arpego Network, Brest, France.22 University Cancer Center Leipzig, Leipzig, Germany. 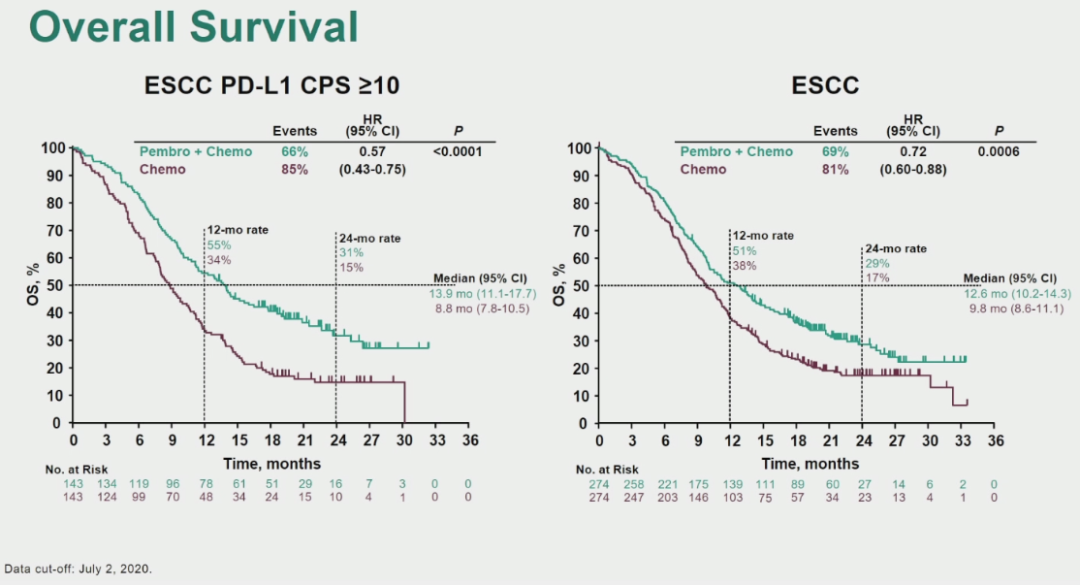
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
